|
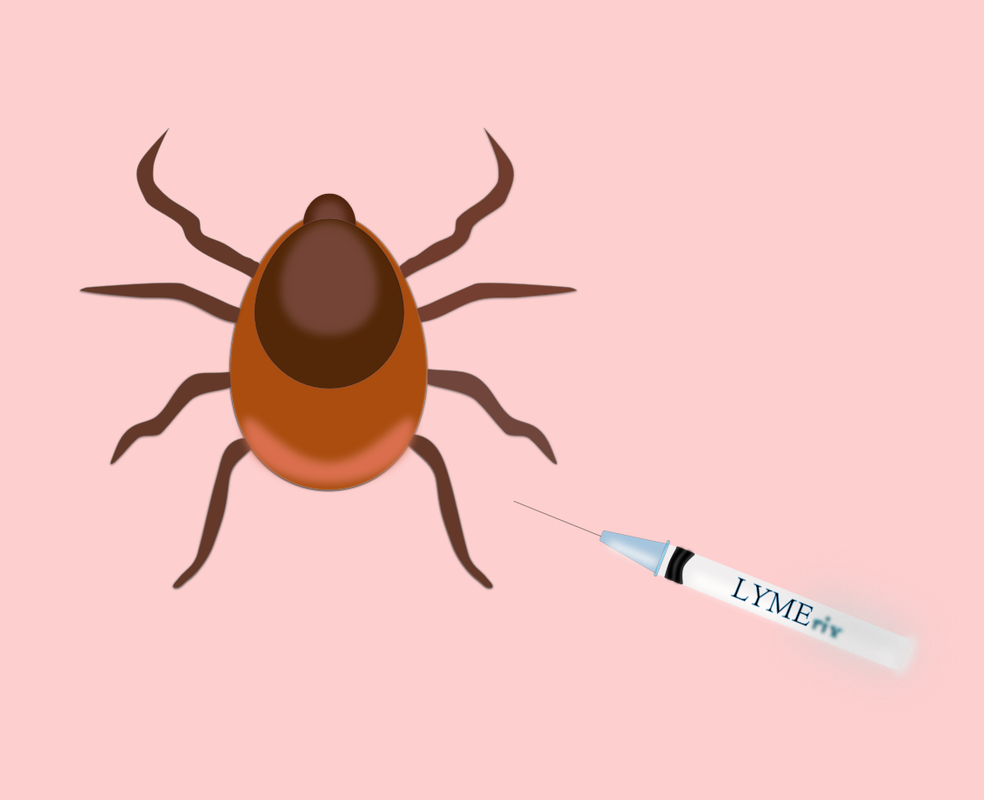
By Zoë Heller How did an 80% effective vaccine get voluntarily removed from the market in only three years? The answer is anti-vaxxers. Lyme disease is an infection caused by the bacterium Borrelia burgdorferi, which is transmitted through black-legged tick bites. While most know to check for the characteristic bulls-eye rash that comes with tick transmission, few know of the devastating, long-term effects that manifest if left untreated. At first, symptoms include fever, fatigue, and headache, but without antibiotics, the infection can spread to the joints, heart, and nervous system. The spreading of the bacterium can cause arthritis, heart palpitations, dizziness, nerve pain, and even inflammation of the brain and spinal cord. With treatment, Lyme disease often goes away; however, a small percentage of people develop what is known as “chronic Lyme disease” or post-treatment Lyme disease syndrome, which results in muscle aches and fatigue that can last for more than 6 months after antibiotic treatment. Unfortunately, Lyme disease infects as many as 300,000 people across the United States each year. The disease is also becoming more common due to climate change, which results in a longer tick season. Currently, the methods for preventing the disease—insect repellant and vigilance—are unreliable. This was untrue in the period from 1998 to 2001, however, when an effective and safe vaccine for Lyme disease—known as LYMErix—was introduced. LYMErix is now known as a cautionary tale among vaccine developers and manufacturers alike. The vaccine was not without its issues, as it required three doses over a year and was not approved for children under 15, but it was up to 90% effective at neutralizing the bacterium before it was even transmitted by the tick. The fatal flaw of LYMErix was its release date—1998. That same year, The Lancet published a study (falsely) claiming that the MMR vaccine was related to autism, thus starting the current anti-vaccination movement. When members of the United States Food and Drug Administration (FDA) panel that unanimously approved LYMErix theorized that the vaccine could cause an autoimmune response in the body—which was later proven to be untrue—the media capitalized on pre-existing vaccine panic, and fear erupted in the public. ABC News and other major outlets reported on vaccine “victims” claiming to have contracted arthritis through LYMErix, though when the FDA investigated these claims, they found no link between the vaccine and joint pain. Major websites such as The Lyme Disease Network devoted coverage to the controversy, which continued to fan the flames of chaos. Eventually, the sensationalized fear of vaccines spawned lawsuits against LYMErix’s manufacturer, GlaxoSmithKline. A class action lawsuit led by 121 “victims” of the vaccine argued that GlaxoSmithKline intentionally concealed evidence of the vaccine’s harmful or adverse effects. No studies confirmed any of these claims, but the damage was already done. The market for a Lyme disease vaccine was already niche, and sales plummeted due to the media’s portrayal of the vaccine. GlaxoSmithKline pulled LYMErix from the market on February 26, 2002. Sources: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2870557/ https://www.vox.com/science-and-health/2018/5/7/17314716/lyme-disease-vaccine-history-effectiveness https://www.cdc.gov/lyme/index.html https://www.vox.com/2018/5/2/17303516/lyme-disease-symptoms-treatment-test https://www.rxlist.com/lymerix-side-effects-drug-center.htm
1 Comment
Thank you for sharing this very informative content I read a lot of importance's to live longer by taking self care by drinking food supplements daily. I also recommend Haarlem Oil made from France. It can help to boost your immune system to fight against viruses and infections.
Reply
Leave a Reply. |
Categories
All
Archives
April 2024
|